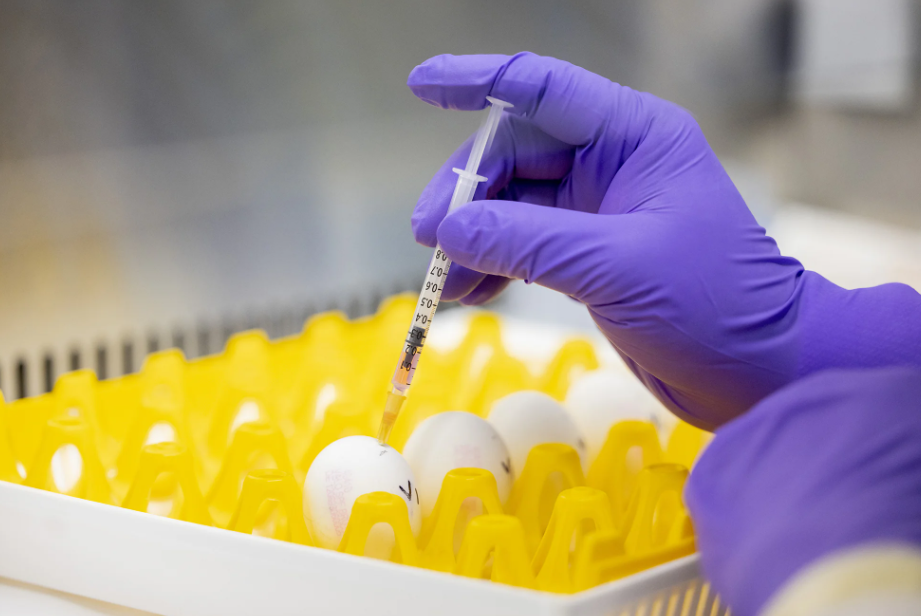
Moderna has begun a late-stage clinical trial of its experimental bird flu vaccine in the United States and the United Kingdom, marking a significant milestone in the effort to prepare for a possible future influenza pandemic. This is the first pandemic bird flu vaccine built with mRNA technology to reach a late-stage trial, moving the company’s candidate into a much more advanced phase of testing than before.
The vaccine, known as mRNA-1018, will be tested in about 4,000 healthy adults aged 18 and older. The study is intended to evaluate the shot in a larger group of participants, which is a key step toward determining whether the vaccine performs well enough to support a future regulatory filing. Late-stage trials matter because they are designed to provide stronger evidence about how well a vaccine works and how safe it appears in broader populations.
The project is also receiving outside support. Trial is backed by a $54 million investment from the Coalition for Epidemic Preparedness Innovations (CEPI), an international partnership focused on speeding the development of vaccines against epidemic and pandemic threats. That funding is especially notable because Moderna had previously lost major U.S. government backing for the same area of work. CEPI’s investment came after U.S. Health Secretary Robert F. Kennedy Jr. canceled more than $700 million in contracts with Moderna in May 2025 that had been intended to support bird flu vaccine development.
The trial highlights the continuing belief in mRNA as a rapid-response vaccine platform. The technology, which became widely known through COVID-19 vaccines, is seen as a faster way to develop shots against emerging viruses with pandemic potential. That speed is one of the main reasons bird flu researchers and preparedness groups are interested in mRNA approaches: if an outbreak worsens or a virus mutates in a dangerous way, a faster vaccine-development system could be crucial.
Moderna is framing the study as part of a broader preparedness strategy. CEO Stephane Bancel said bird flu remains a pandemic threat and described the trial as an important step toward improving readiness for future outbreaks. The company also said that any eventual regulatory filing for mRNA-1018 would be supported by positive data from its separate seasonal flu vaccine candidate, mRNA-1010, which is currently under review in the United States, European Union, Canada, and Australia. That means Moderna appears to be trying to build a broader influenza-vaccine platform rather than treat bird flu as a one-off effort.
There is also a global-access component built into the CEPI agreement. If the bird flu vaccine is approved and a flu pandemic is declared, Moderna has agreed to reserve 20% of its manufacturing capacity for supply to low- and middle-income countries at affordable prices. That provision reflects lessons from the COVID era, when vaccine access was highly unequal across countries.
Overall, the new trial represents an important scientific and public health step. It does not mean a bird flu vaccine is ready for use yet, but it does show that pandemic-preparedness efforts are moving forward despite political and funding setbacks. By advancing mRNA-1018 into late-stage testing, Moderna is trying to prove that mRNA can be used not only for COVID and seasonal flu, but also for future influenza threats that could emerge with far less warning.